 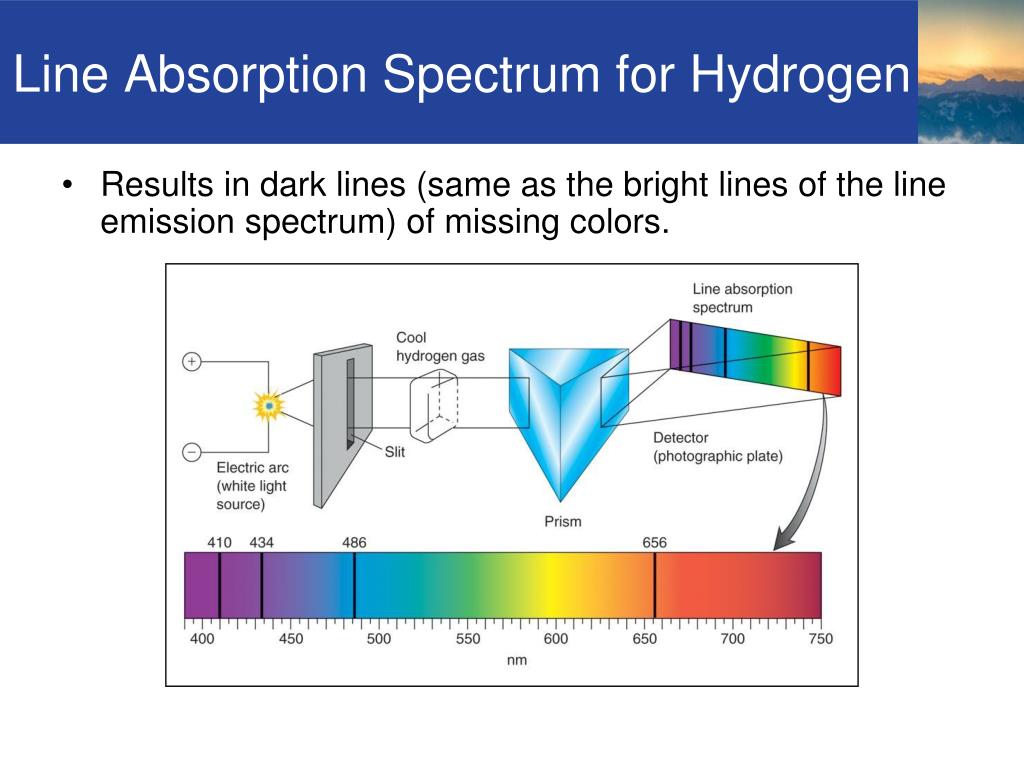
The range of forbidden energies as determined by the spectroscopic method is found to be 1.0 ev and 0.8 ev for Si and Ge, respectively. Atoms and molecules can absorb and emit light, telling us how. We can see emission spectra from comets, nebula and certain types of stars. As with absorption spectra, the pattern of these lines is unique for each element. With the aid of the present measurements and Skinner's data on the L emission bands of Si, it has been possible to arrive at an estimation of the magnitude of the energy gap in each semiconductor on the basis of the observed interval between emission and absorption edges. An emission spectrum occurs when the atoms and molecules in a hot gas emit light at certain wavelengths, causing bright lines to appear in a spectrum. Two bands, whose peaks fall at 102.4 A and 104.3 A, have been identified as the M 2 and M 3 emission spectra, corresponding to transitions from the valence band into the M 2 and M 3 levels. 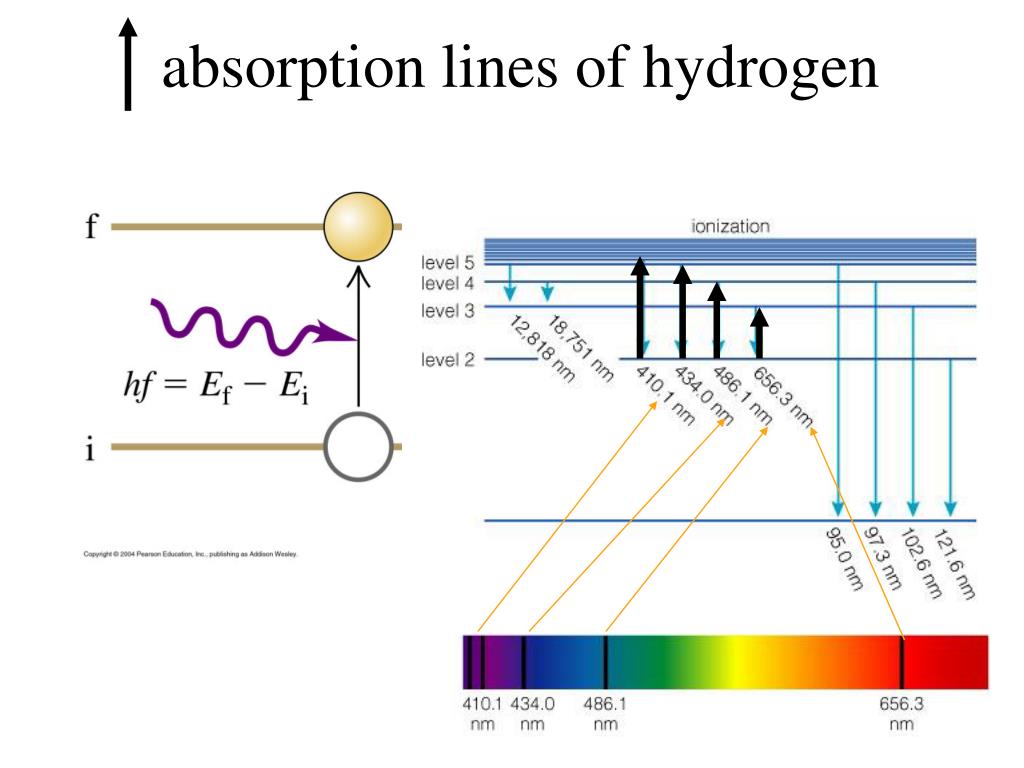
The emission spectrum of a Ge target was also studied in the 60 A to 600 A region. The linear absorption coefficients on the high-energy side of the respective edges of Si and Ge are 1.4× 10 5 cm − 1 and 1.8× 10 5 cm − 1. The L 2, 3 edge of Si appears at 123 A, while the M 2, 3 edge of Ge is centered at 101.5 A. The spectral region covers the L 2, 3 and M 2, 3 absorption discontinuities of Si and Ge, respectively. The thicknesses of the various absorbers ranged from several hundred to several thousand angstroms. absorption spectra always contain the same wavelength ranges c) The emission spectrum contains the same exact wavelengths as the absorption spectrum d) The. A demonstration of the 589 nm D 2 (left) and 590 nm D 1 (right) emission sodium D lines using a wick with salt water in a flame. The emission spectrum of a fluorophore is the image of its absorption spectrum when the probability of the S1 S0 transition is identical. Experimental and simulated absorption and emission spectra of pyrene have been previously reported by several research groups. 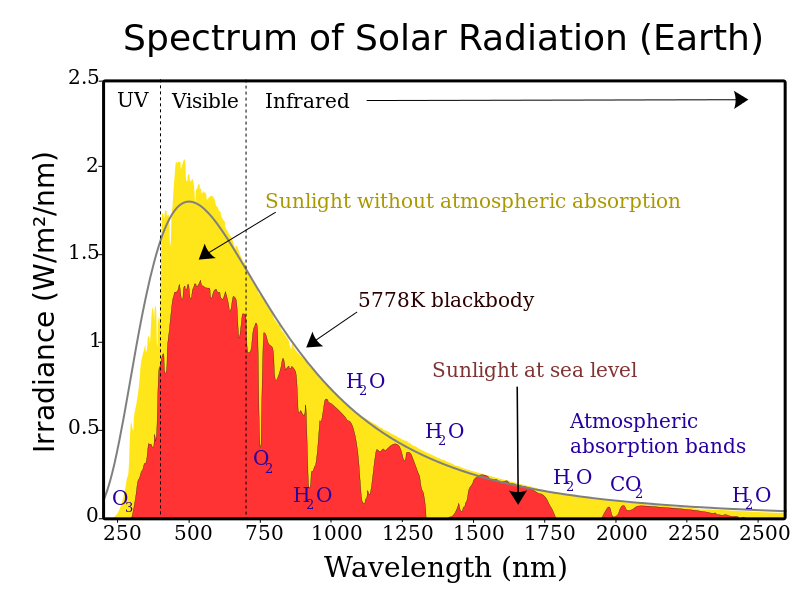
76 They obtained a large red shift of the vertical. The set of wavelengths that a pigment doesn't absorb are reflected, and the reflected light is what we see as color. When an atom gives an absorption spectrum, it is because it has gained a higher energy level. An absorption spectrum is characterized by dark lines or gaps, while an emission spectrum typically shows colored lines. But the emission spectrum is a result of atoms releasing energy. In the diagram below, you can see the absorption spectra of three key pigments in photosynthesis: chlorophyll a, chlorophyll b, and -carotene. An absorption spectrum is observed when atoms absorb some energy. Emission Spectrum: Starlight can also heat up a cloud of gas, exciting the atoms and molecules within the gas, and causing it to emit light. The absorption spectrum of pentacene calculated at the GGA level has recently been reported. The set of wavelengths absorbed by a pigment is its absorption spectrum. The absorption spectra of thin evaporated foils of Si and Ge have been investigated in the soft x-ray region extending from 70 A to 200 A. Emission spectrum of a ceramic metal halide lamp. An absorption spectrum has dark lines or gaps in the spectrum corresponding to wavelengths that are absorbed by the gas. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
